| 10/19/2025 | 10:45 - 11:30 am | PW07 – Expanding Your Influence to Improve Antibiotic Use in Outpatient Settings | Advancing Outpatient Stewardship in Health Systems | | Ritu Banerjee |
| 10/19/2025 | 10:50 am - 12:15 pm | PW04 – Fellows’ Day Workshop | Careers in Research | | Cesar Arias |
| 10/19/2025 | 2:40 – 3:15 pm | 2 – Beyond the 101: Pondering Perplexing Prescribing Issues | Can We Use Ceftriaxone Susceptibility to Interpret Susceptibility to Third-Generation Cephalosporins? | | Pranita Tamma |
| 10/19/2025 | 3:45 – 5:30 pm | 8 – Opening Plenary: Reflection and Renewal: Advancing Public Health in Challenging Times | | Yohei Doi | |
| 10/20/2025 | 8:00 – 9:00 am | 15 – New Antimicrobials in the Pipeline | | David van Duin | |
| 10/20/2025 | 8:00 – 8:30 am | 17 – These Blind Mice: When Bench Doesn’t Meet Bedside | Optimizing Dosing Strategies: Insights From Systems Pharmacology and Experimental Models | | Gauri Rao |
| 10/20/2025 | 8:00 – 9:00 am | 18 – Top 10 Papers in Antimicrobial Resistance | | Yohei Doi | |
| 10/20/2025 | 10:55 - 11:20 am | 24 – Applying Next-Generation Sequencing to Clinical Treatment and Prevention | Cases in Genotype-Phenotype in the Microbiology Laboratory: What Clinician Need to Know | | Amy Mathers |
| 10/20/2025 | 10:30 - 11:45 am | 25 – Blood and Guts: Microbiome Discoveries | | David van Duin | |
| 10/20/2025 | 10:30 - 11:45 am | 30 – Lessons Learned in Biopreparedness and Biocontainment: Lassa Fever in the Midwest | | Yohei Doi | |
| 10/20/2025 | 2:21 – 2:39 pm | 64 – Out With the Old, in With the New: Time to Update the Management of Staphylococcus aureus infections | Should Ceftobiprole Replace Ceftaroline in the Management of Persistent Staphylococcal Bacteremia? | | Nicholas Turner |
| 10/20/2025 | 1:45 – 3:00 pm | 72 – Late Breaking Trial Results for Viral and Bacterial Infections | | Yohei Doi | |
| 10/20/2025 | 3:15 – 4:30 pm | 84 – Diagnostic Clinical Cases: What Do I Do With This Test Result? | | Ritu Banerjee | |
| 10/20/2025 | 3:15-4:30 pm | 87 – Extra, Extra New Info on Treating and Preventing RSV and Influenza | | Angelique Boutzoukas | |
| 10/20/2025 | 3:15-4:30 pm | 88 – New Anti-Infective Agents | | Pranita Tamma | |
| 10/20/2025 | 3:15-4:30 pm | 84 – Commonalities and Differences in Antimicrobial Treatment of Resistant Pathogens | | Yohei Doi | |
| 10/21/2025 | 8:00 – 9:00 am | 99 – Diagnostic Trials: Past and Future | | Robin Patel, Ritu Banerjee | |
| 10/21/2025 | 8:00 – 8:30 am | 99 – Diagnostic Trials: Past and Future | Evaluation of Infectious Disease Diagnostics: Innovative Study Designs to Assess Test Performance | | Michael Satlin |
| 10/21/2025 | 8:30 – 9:00 am | 99 – Diagnostic Trials: Past and Future | Evaluation of Infectious Disease Diagnostics: Study Designs to Assess Impact on Clinical Outcomes | | Sarah Doernberg |
| 10/21/2025 | 8:00 – 9:00 am | 104 – Pediatric Antimicrobial Resistance: Key Updates for the Busy Clinician | | Latania Logan, Pranita Tamma | |
| 10/21/2025 | 8:30 – 9:00 am | 104 – Pediatric Antimicrobial Resistance: Key Updates for the Busy Clinician | Updates in Gram-Negative Bacteria | | Angelique Boutzoukas |
| 10/21/2025 | 9:15 -10:00 am | 107 – Maxwell Finland Lecture | | Yohei Doi | |
| 10/21/2025 | 9:35 -10:00 am | 107 – Maxwell Finland Lecture | Antimicrobial Resistance at the Bedside: Translating Science into Patient Outcomes | | Cesar Arias |
| 10/21/2025 | 1:45 – 3:00 pm | 157 – Cavities, Appys, and CLABSis, Oh My! Perspectives From Our Surgery and Radiology Colleagues | | Latania Logan | |
| 10/21/2025 | 1:45 – 3:00 pm | 162 – Fresh New Results On Old and New Vaccines | | Ahmed Babiker | |
| 10/21/2025 | 1:45 – 3:00 pm | 163 – Lowering Barriers to Diagnostics Access to Reduce Health Inequalities and Improve Outcomes | | Ritu Banerjee | |
| 10/21/2025 | 3:15 – 4:30 pm | 171 – New Antibiotics, Untapped Potential: Exploring the Underuse of Recently Approved Agents | | Thomas Lodise | |
| 10/21/2025 | 4:05 – 4:30 pm | 172 – Bug to Drug: Translating Mechanisms of Antimicrobial Resistance to Treatment of Challenging Infections | Translational Antimicrobial Resistance Research in Low-Resource Settings | | Jose Munita |
| 10/21/2025 | 3:15 – 4:30 pm | 173 – Challenging Cases in Immunocompromised Host | | | Michael Satlin (panelist) |
| 10/21/2025 | 5:25 – 5:55 pm | 183 – SHEA Lectureship | Where is the True North of Infection Control? | | Anthony Harris |
| 10/21/2025 | 4:45 – 6:00 pm | 184 – Stanley A. Plotkin Lecture in Vaccinology | | Latania Logan | |
| 10/22/2025 | 8:00 –8:20 am | 191 – One Size Fits One: Toward Individualized Management of Infectious Diseases | Molecular Diagnostics to Refine Duration of Therapy for Blood Stream Infection: S. aureus | | Vance Fowler |
| 10/22/2025 | 10:30 -10:55 am | 197 – Clinical Trials That Will Change Your Practice | Trials in Bacteriology That May Change Your Practice | | Amy Mathers |
| 10/22/2025 | 1:45-3:00 pm | 241 – Advances in Managing Antimicrobial Toxicities | | Amit Pai | |
| 10/22/2025 | 3:15 – 4:30 pm | 257 – Cracking the Code of Resistance: A Deep Dive Into Metallo-Beta-Lactamase From Bench to Bedside | | Yohei Doi, Amy Mathers | |
| 10/22/2025 | 3:15 – 3:40 pm | 257 – Cracking the Code of Resistance: A Deep Dive Into Metallo-Beta-Lactamase From Bench to Bedside | Unmasking Resistance: Epidemiology and Diversity of MBLs | | Maria Mojica |
| 10/22/2025 | 3:15 – 4:30 pm | 259 – Driving Better Decisions: Incentivizing Diagnostic Stewardship | | Ritu Banerjee | |
| 10/22/2025 | 3:15 – 4:30 pm | 266 – Why Do We Do This? Exploring the Data Behind Long-Established Practices in ID | Double Coverage; Do We Need It? | | Kerry LaPlante |


 The results of an ARLG study, Dalbavancin as an Option for Treatment of S. aureus Bacteremia (DOTS), were recently published in
The results of an ARLG study, Dalbavancin as an Option for Treatment of S. aureus Bacteremia (DOTS), were recently published in 
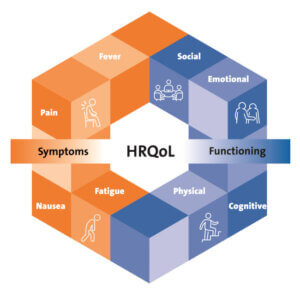 The ARLG published a new supplement in the journal of Clinical Infectious Diseases titled “In Pursuit of Patient-Centered Antibacterial Trials: Listening to, Learning From, and Leveraging the Patient Perspective.” The supplement includes eight articles authored by ARLG members that describe several challenges and opportunities for measuring health-related quality of life (HRQoL) in patient care and in clinical studies of infectious diseases.
The ARLG published a new supplement in the journal of Clinical Infectious Diseases titled “In Pursuit of Patient-Centered Antibacterial Trials: Listening to, Learning From, and Leveraging the Patient Perspective.” The supplement includes eight articles authored by ARLG members that describe several challenges and opportunities for measuring health-related quality of life (HRQoL) in patient care and in clinical studies of infectious diseases.
